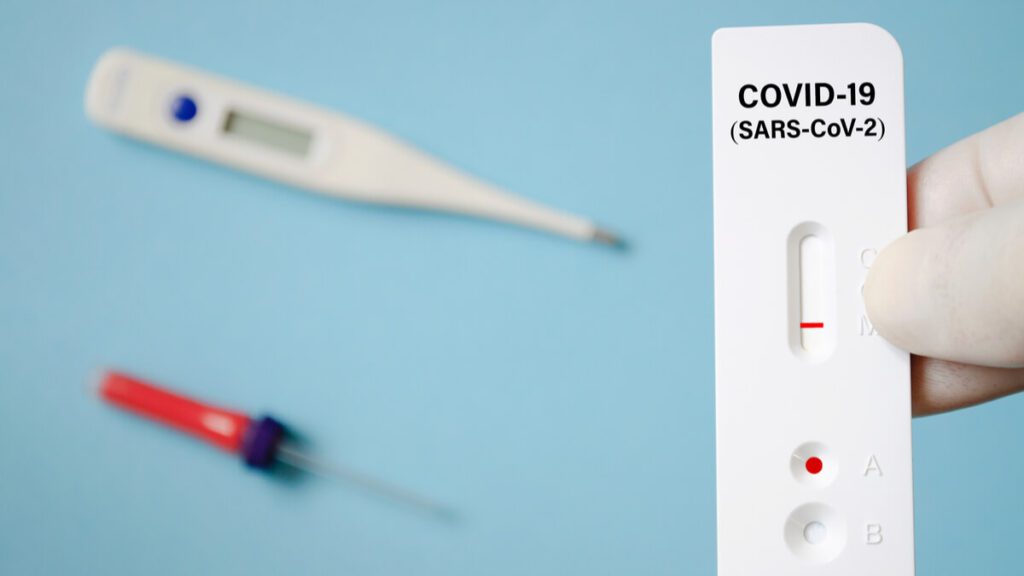
The Food and Drug administration of the United Sates authorized the emergency use of the first COVID-19 antigen test to detect the molecules responsible for stimulating an immune response against COVID-19.
The test is a fast and efficient way to test for antigens in a similar fashion to a pregnancy test – but using oral and nasal swabs instead of urine. After the swab is placed on the testing card, in 15 minutes the card reveals either one line meaning the absence of antigens, or two lines meaning the presence of antigens.
“This new COVID-19 antigen test is an important addition to available tests because the results can be read in minutes, right off the testing card. This means people will know if they have the virus in almost real-time”. Says Jeff Shuren, MD, JD, and Director of Center for Devices and Radiological Health, Food and Drug Administration. “Due to its simpler design and the large number of tests the company anticipates making in the coming months, this new antigen test is an important advancement in our fight against the pandemic.”
Given the simplicity of the COVID-19 antigen test, Abbott, the manufacturer of the test, expects there to be 50 million made available on a monthly basis in the US as of October. This would greatly bolster the U.S testing capabilities, and make easy and fast testing available to as many people as needed.
However, the current COVID-19 antigen test lacks some degree of sensitivity than the well known PCR molecular test. Therefore, tests that results in a negative should be taken contextually, and through the physician’s observations of symptoms, as well as patient medical history, and verified with a PCR test.
Source:
