New device to help protect athletes from traumatic brain injury
A new device that could help reduce the risk of traumatic brain injury during head impacts was given approval this week by the Food and Drug Administration. The device is authorized for athletes 13 and older, and can be used during football, soccer and other high-impact sports.
The device is intended to protect athletes from sub concussive injuries, by protecting the internal jugular veins, thereby increasing the volume of blood to vessels of the skull.
The additional blood restricts the movement of the brain inside the skull. As the brain moves less, it may be protected from the negative effects of the “slosh” movement that occurs in unprotected athletes who suffer heavy impacts to the head.
The C-shaped device, called the Q-Collar, fits around the back and side of the neck. It works by clamping compressive protection on blood vessels in the neck.
A CDC (Centers for Disease Control and Prevention) analysis found 2 million children visited emergency departments because of TBI sustained during sports and recreation activities from 2010 to 2016.
TBI can have emotional, physiological and cognitive effects, while negative health outcomes can stem from sub concussive injuries that alter tissue but do not cause diagnosed concussion.
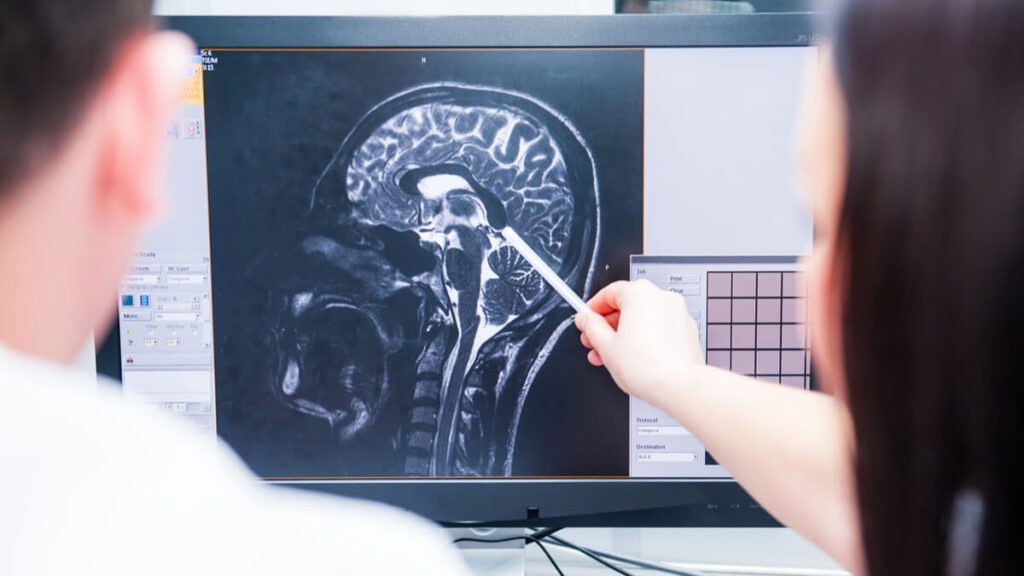
The researchers used advanced imaging techniques to look at changes in the brains of nearly 300 study participants before and after the season. They found changes in deep structures of the brain in 73 percent of participants in the no-collar group, while no significant changes in these same structures were found in 77 percent of participants in the collar group.
Carolina Panthers’ Linebacker Luke Kuechly, who retired at 28 after suffering a series of head injuries, was seen wearing the Q-Collar in his final seasons with the NFL.
“Today’s action provides an additional piece of protective equipment athletes can wear when playing sports to help protect their brains from the effects of repetitive head impacts while still wearing the personal protective equipment associated with the sport,” said Dr. Christopher M. Loftus, acting director of the Office of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health.
